Toronto, CANADA
Navient Cranial Navigation System receives CE Mark
Navient brings Simplicity, Accuracy and Affordability to Cranial Navigation Systems.
ClaroNav Kolahi Inc. (CKI) announced today that it has received CE Mark approval for the commercial sale of its Navient Cranial navigation system in Europe.
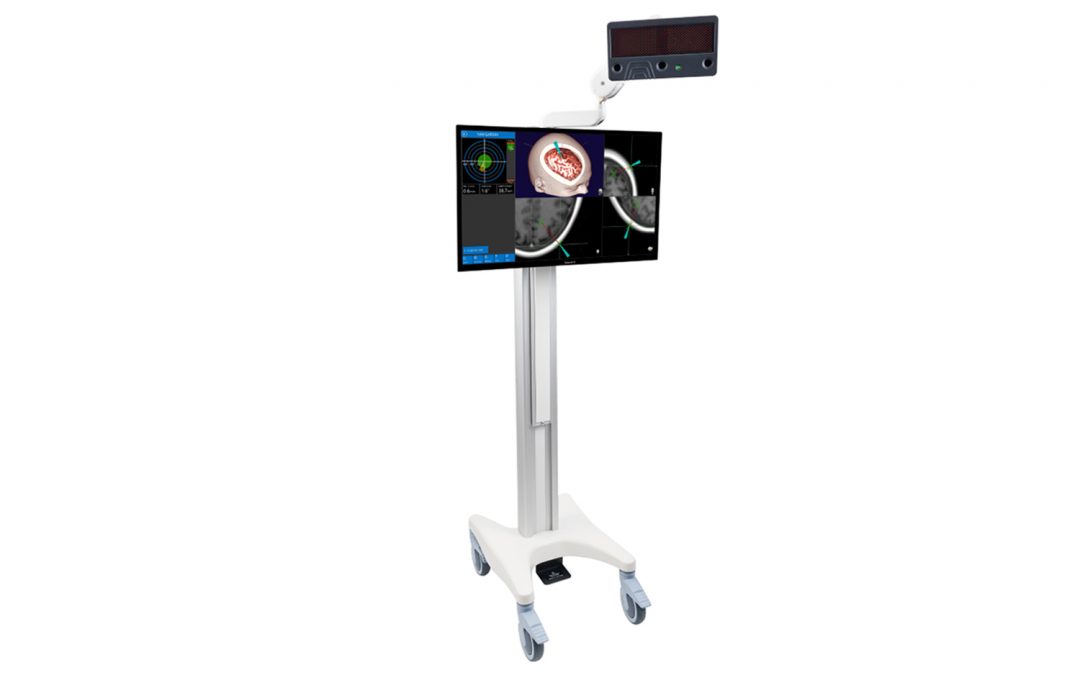
Navient is a computerized surgical navigation system intended as an aid for precisely locating anatomical structures in either open or percutaneous procedures. The Navient system is indicated for any cranial interventions in which the use of stereotactic surgery may be appropriate.
Navient utilizes the latest technologies in surgical navigation systems and brings simplicity, accuracy and affordability to the cranial navigation system. It offers truly portable cart along with powerful and intuitive software. Its instruments are steam sterilizable, and there is no consumable cost per procedure.
For more information about the Navient, please visit https://www.claronav.com/navient/
About ClaroNav Kolahi Inc. (CKI)
CKI is a medical device hardware and software company headquartered in Toronto, Canada, and is represented worldwide by authorized distributors. CKI is deeply committed to product quality and safety, and certified as compliant with ISO 13485, ISO 14971 and CMDCAS. CKI is dedicated to innovation and developing state-of-the-art surgical navigation systems to enable surgeons to confidently make more informed decisions for better patient outcomes.
For more information, visit www.claronav.com
Contact
ClaroNav Kolahi Inc.
1140 Shepard Avenue West
Toronto, Canada, M3K 2A2
Phone: + 1 (647) 951 1525
Website: www.claronav.com
Email: [email protected]